|
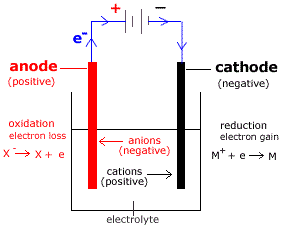
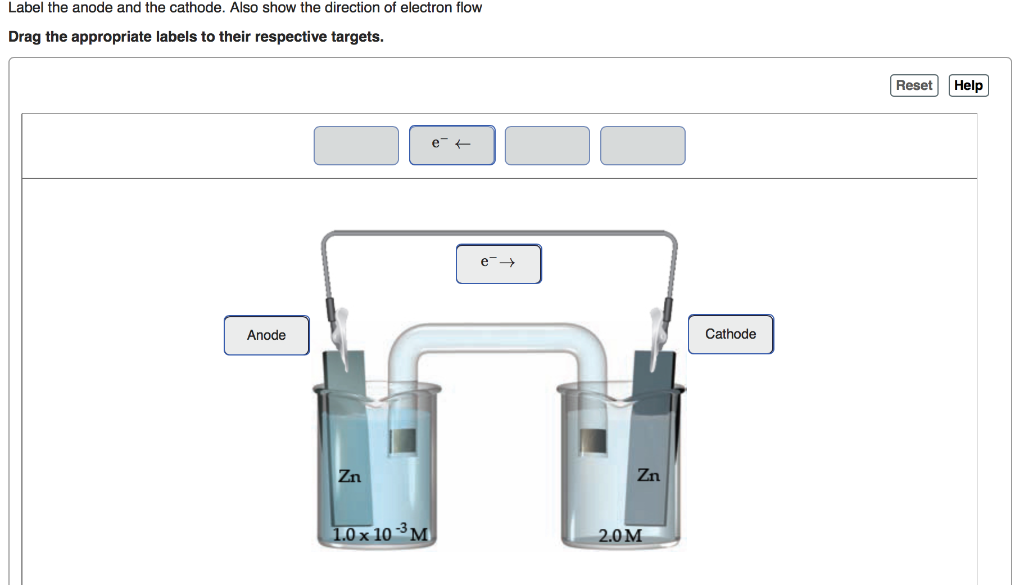
1/1/2024 0 Comments Anode cathodeFor a galvanic cell, anode holds the negative polarity whereas cathode acts as a positive terminal.Thus, an anode attracts negatively charged particles while a cathode attracts positively charged particles. For an electrolytic cell, anode acts as a positive terminal while a cathode holds negative polarity.This is so because the terminal polarity varies according to the type of cell i.e., electrolytic or galvanic in use. The specific denotation of the anode as positive and cathode as negative is wrong.While cathode corresponds to the electrode where reduction i.e., the gain of electrons occurs. The key factor of differentiation between anode and cathode is that anode corresponds to the electrode where oxidation i.e., loss of electrons occurs.Key Differences Between Anode and Cathode In electrolytic cell, it is a source of negative charge or electron donor. In electrolytic cell it is a source of positive charge or electron acceptor. Content: Anode Vs CathodeĪn anode in electrolytic cell attracts anions.Ī cathode in electrolytic cell attracts cations. when the electrode gains electrons which are released by the electrolyte undergoes reduction. However, when the reverse operation occurs i.e. It specifies the conductive phase where the transfer of charged carriers takes place.Īn electrode that loses electrons and is accepted by the electrolyte undergoes oxidation. More specifically, we can say, it is regarded as a surface where a redox reaction takes place between the metal and the solution.Įlectrode generally represents an electrical conductor/ semiconductor within the electrochemical cell. Electrode acts as a metallic contact through which the current enters and leaves the electrolyte. But first, see-Ī crucial component of an electrochemical cell that makes contact with electrolyte is known as an electrode. But in this content, you will get to know that the differentiation between anode and cathode is not merely done according to the type of polarity. That is all I have to share about the said topic.People generally in misconception regards anode particularly as positive and cathode particularly as negative. In this article, we just try to talk about electrolytic capacitor anode cathode identification. you have to identify its anode and cathode before placing it in any circuit. polarity dependent.įor the electrolytic capacitor, polarity is important, i.e. 
It has two types, ceramic which is nonpolar, and the electrolytic capacitor which is polar, i.e. ConclusionĬapacitors are the most used component in electrical and electronics circuits. Just simply put the capacitor in the tester, press test, and get the results. It doesn’t matter for this method what is the length of any leg of your given capacitor. On the screen, you will see the circuit symbol of your capacitor with its right pin configuration.Īlso, this method is applicable to both old and new capacitors.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
